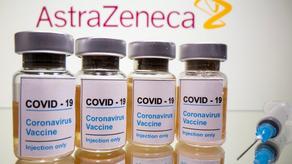
The EU’s drug watchdog said on Thursday it is still convinced the benefits of AstraZeneca’s COVID-19 vaccine outweigh the risks following an investigation into reports of blood disorders that prompted more than a dozen nations to suspend its use.
The news came as the European Medicines Agency (EMA) director Emer Cooke said the watchdog could not definitively rule out a link between blood clot incidents and the vaccine in its investigation into 30 cases of a rare blood clotting condition.
But she said in a briefing the “clear” conclusion of the review was that the vaccine “benefits in protecting people from COVID-19 with the associated risk of death or hospitalisation outweighs the possible risks.”
The agency will however update its guidance to include an explanation about the potential risks on both the patient leaflet and in the information for healthcare professionals, she said.
“This is a safe and effective vaccine,” said Cooke.
The agency has been under growing pressure to clear up safety concerns after a small number of reports in recent weeks of bleeding, blood clots and low platelet counts in people who have received the shot.