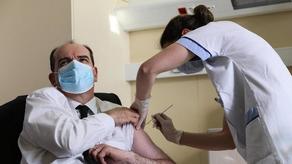
French Prime Minister Jean Castex, 55, received the jab at the Begin military hospital near Paris as France's medical regulator Haute Autorité de Santé ruled on Friday that use of the AstraZeneca vaccine in France could resume after a suspension of several days.
Vaccination with AstraZeneca has resumed in France and Germany today. The decision to re-use the drug has been made by Italy, Cyprus, Spain, Latvia, and Lithuania.
The recently published WHO statement says that deep venous thrombosis or pulmonary embolism cannot be caused by the AstraZeneca vaccine, which remains an important tool against the coronavirus.
"Based on a careful scientific review of the available information, the subcommittee came to the following conclusions and recommendations: the AstraZeneca COVID-19 vaccine (including Covishield) continues to have a positive benefit-risk profile, with tremendous potential to prevent infections and reduce deaths across the world. The available data do not suggest any overall increase in clotting conditions such as deep venous thrombosis or pulmonary embolism following administration of COVID-19 vaccines. Reported rates of thromboembolic events after COVID-19 vaccines are in line with the expected number of diagnoses of these conditions. Both conditions occur naturally and are not uncommon. They also occur as a result of COVID-19," the statement reads.
The EU’s drug watchdog said on Thursday it is still convinced the benefits of AstraZeneca’s COVID-19 vaccine outweigh the risks following an investigation into reports of blood disorders that prompted more than a dozen nations to suspend its use.